Abstract
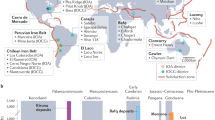
In situ rover investigations on Mars have discovered manganese oxides as fracture-filling materials at Gale and Endeavour craters. Previous studies interpreted these minerals as indicators of atmospheric oxygen on early Mars. By contrast, we propose that the oxidation of manganese by oxygen is highly unlikely because of exceedingly slow reaction kinetics under Mars-like conditions and therefore requires more reactive oxidants. Here we conduct kinetic experiments to determine the reactivity of the oxyhalogen species chlorate and bromate for oxidizing dissolved Mn(ii) in Mars-like fluids. We find that oxyhalogen species, which are widespread on the surface of Mars, induce substantially greater manganese oxidation rates than O2. From comparisons of the potential oxidation rates of all available oxidants (including reactive oxygen species peroxide and superoxide), we suggest that the oxyhalogen species are the most plausible manganese oxidants on Mars. In addition, our experiments precipitated the manganese oxide mineral nsutite, which is spectrally similar to the dark manganese accumulations reported on Mars. Our results provide a feasible pathway to form manganese oxides under expected geochemical conditions on early Mars and suggest that these phases may record an active halogen cycle rather than substantial atmospheric oxygenation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data associated with the manuscript are available at: https://figshare.com/s/e12d62da416302225cf3.
Code availability
The code to model Mn(ii) oxidation by O2 in the Geochemist’s Workbench is available at https://figshare.com/articles/online_resource/MnII_oxidation_species_Morgan_V7_rea/21066232.
References
Lanza, N. L. et al. High manganese concentrations in rocks at Gale crater, Mars. Geophys. Res. Lett. 41, 5755–5763 (2014).
Lanza, N. L. et al. Oxidation of manganese in an ancient aquifer, Kimberley formation, Gale crater, Mars. Geophys. Res. Lett. 43, 7398–7407 (2016).
Berger, J. A. et al. Elemental composition and chemical evolution of geologic materials in Gale crater, Mars: APXS results from Bradbury landing to the Vera Rubin ridge. J. Geophys. Res. Planets 125, e2020JE006536 (2020).
Arvidson, R. E. et al. High concentrations of manganese and sulfur in deposits on Murray Ridge, Endeavour crater, Mars. Am. Mineral. 101, 1389–1405 (2016).
Mittlefehldt, D. W. et al. Diverse lithologies and alteration events on the rim of Noachian‐aged Endeavour crater, Meridiani Planum, Mars: in situ compositional evidence. J. Geophys. Res. Planets 123, 1255–1306 (2018).
Wordsworth, R. et al. A coupled model of episodic warming, oxidation and geochemical transitions on early Mars. Nat. Geosci. 14, 127–132 (2021).
Liu, Y. et al. Manganese oxides in Martian meteorites Northwest Africa (NWA) 7034 and 7533. Icarus 364, 114471 (2021).
Diem, D. & Stumm, W. Is dissolved Mn2+ being oxidized by O2 in absence of Mn-bacteria or surface catalysts? Geochim. Cosmochim. Acta 48, 1571–1573 (1984).
Trainer, M. G. et al. Seasonal variations in atmospheric composition as measured in Gale crater, Mars. J. Geophys. Res. Planets 124, 3000–3024 (2019).
Davies, S. H. & Morgan, J. J. Manganese (ii) oxidation kinetics on metal oxide surfaces. J. Colloid Interface Sci. 129, 63–77 (1989).
Morgan, J. J. Kinetics of reaction between O2 and Mn(ii) species in aqueous solutions. Geochim. Cosmochim. Acta 69, 35–48 (2005).
von Langen, P. J., Johnson, K. S., Coale, K. H. & Elrod, V. A. Oxidation kinetics of manganese(ii) in seawater at nanomolar concentrations. Geochim. Cosmochim. Acta 61, 4945–4954 (1997).
Madden, A. S. & Hochella, M. F. A test of geochemical reactivity as a function of mineral size: manganese oxidation promoted by hematite nanoparticles. Geochim. Cosmochim. Acta 69, 389–398 (2005).
Baron, F., Gaudin, A., Lorand, J. P. & Mangold, N. New constraints on early Mars weathering conditions from an experimental approach on crust simulants. J. Geophys. Res. Planets 124, 1783–1801 (2019).
Kite, E. S. & Daswani, M. M. Geochemistry constrains global hydrology on early Mars. Earth Planet. Sci. Lett. 524, 115718 (2019).
Wang, X. et al. The presence of ferrihydrite promotes abiotic formation of manganese (oxyhydr)oxides. Soil Sci. Soc. Am. J. 79, 1297–1305 (2015).
Lan, S. et al. Mechanisms of Mn(ii) catalytic oxidation on ferrihydrite surfaces and the formation of manganese (oxyhydr) oxides. Geochim. Cosmochim. Acta 211, 79–96 (2017).
Treiman, A. H. et al. Mineralogy, provenance, and diagenesis of a potassic basaltic sandstone on Mars: CheMin X‐ray diffraction of the Windjana sample (Kimberley area, Gale crater). J. Geophys. Res. Planets 121, 75–106 (2016).
Zhao, Y. Y. S., McLennan, S. M., Jackson, W. A. & Karunatillake, S. Photochemical controls on chlorine and bromine geochemistry at the Martian surface. Earth Planet. Sci. Lett. 497, 102–112 (2018).
Sutter, B. et al. Measurements of oxychlorine species on Mars. Int. J. Astrobiol. 16, 203–217 (2017).
Bellucci, J. et al. Halogen and Cl isotopic systematics in Martian phosphates: implications for the Cl cycle and surface halogen reservoirs on Mars. Earth Planet. Sci. Lett. 458, 192–202 (2017).
Mitra, K. & Catalano, J. G. Chlorate as a potential oxidant on Mars: rates and products of dissolved Fe(ii) oxidation. J. Geophys. Res. Planets 124, 2893-2916 (2019).
Mitra, K., Moreland, E. L. & Catalano, J. G. Capacity of chlorate to oxidize ferrous iron: implications for iron oxide formation on Mars. Minerals 10, 729 (2020).
Mitra, K., Moreland, E. L., Knight, A. L. & Catalano, J. G. Rates and Products of Iron Oxidation by Chlorate at Low Temperatures (0 to 25 °C) and Implications for Mars Geochemistry. ACS Earth Space Chem. 6, 250–260 (2022).
Zheng, D., Yin, Z., Zhang, W., Tan, X. & Sun, S. Novel branched γ-MnOOH and β-MnO2 multipod nanostructures. Cryst. Growth Des. 6, 1733–1735 (2006).
Dreibus, G. & Wanke, H. Mars, a volatile-rich planet. Meteoritics 20, 367–381 (1985).
Rampe, E. B., Cartwright, J. A., McCubbin, F. M. & Osterloo, M. M. in D.E. Harlov, L. Aranovich (eds.) The Role of Halogens in Terrestrial and Extraterrestrial Geochemical Processes 959–995 (Springer, 2018).
VanBommel, S., Gellert, R., Berger, J., Yen, A. & Boyd, N. Mars science laboratory alpha particle X-ray spectrometer trace elements: situational sensitivity to Co, Ni, Cu, Zn, Ga, Ge, and Br. Acta Astronaut. 165, 32–42 (2019).
Wang, X. et al. Multiphase volatilization of halogens at the soil–atmosphere interface on Mars. J. Geophys. Res. Planets 126, e2021JE006929 (2021).
Thompson, R. C. Reduction of bromine(v) by cerium(iii), manganese(ii), and neptunium(v) in aqueous sulfuric acid. J. Am. Chem. Soc. 93, 7315–7315 (1971).
Birk, J. P. Kinetics and mechanism of the reduction of bromate ion by hexachloroiridate(iii). Inorg. Chem. 17, 504–506 (1978).
Le, N. C. & Van Phuc, D. Sorption of lead(ii), cobalt(ii) and copper(ii) ions from aqueous solutions by γ-MnO2 nanostructure. Adv. Nat. Sci. Nanosci. Nanotechnol. 6, 025014 (2015).
Post, J. E. Manganese oxide minerals: crystal structures and economic and environmental significance. Proc. Natl Acad. Sci. USA 96, 3447–3454 (1999).
Noda, N. et al. Highly oxidizing aqueous environments on early Mars inferred from scavenging pattern of trace metals on manganese oxides. J. Geophys. Res. Planets 124, 1282–1295 (2019).
Luther, G. W. The role of one- and two-electron transfer reactions in forming thermodynamically unstable intermediates as barriers in multi-electron redox reactions. Aquat. Geochem. 16, 395–420 (2010).
Hem, J. D. Rates of manganese oxidation in aqueous systems. Geochim. Cosmochim. Acta 45, 1369–1374 (1981).
Coughlin, B. R. & Stone, A. T. Nonreversible adsorption of divalent metal ions (Mnii, Coii, Niii, Cuii, and Pbii) onto goethite: effects of acidification, FeII addition, and picolinic acid addition. Environ. Sci. Technol. 29, 2445–2455 (1995).
Jacobsen, F., Holcman, J. & Sehested, K. Oxidation of manganese(ii) by ozone and reduction of manganese(iii) by hydrogen peroxide in acidic solution. Int. J. Chem. Kinet. 30, 207–214 (1998).
Yen, A., Kim, S., Hecht, M., Frant, M. & Murray, B. Evidence that the reactivity of the Martian soil is due to superoxide ions. Science 289, 1909–1912 (2000).
Zent, A. P., Ichimura, A. S., Quinn, R. C. & Harding, H. K. The formation and stability of the superoxide radical (O2−) on rock‐forming minerals: band gaps, hydroxylation state, and implications for Mars oxidant chemistry. J. Geophys. Res. Planets 113, E09001 (2008).
Learman, D. R., Voelker, B. M., Madden, A. S. & Hansel, C. M. Constraints on superoxide mediated formation of manganese oxides. Front. Microbiol. 4, 262 (2013).
Liu, D. & Kounaves, S. P. The role of titanium dioxide (TiO2) in the production of perchlorate (ClO4–) from chlorite (ClO2–) and chlorate (ClO3–) on Earth and Mars. ACS Earth Space Chem. 3, 1678–1684 (2019).
Rao, B., Anderson, T. A., Redder, A. & Jackson, W. A. Perchlorate formation by ozone oxidation of aqueous chlorine/oxy-chlorine species: role of ClxOy radicals. Environ. Sci. Technol. 44, 2961–2967 (2010).
Hamilton, G., Chiswell, B., Terry, J., Dixon, D. & Sly, L. Filtration and manganese removal. J. Water Supply Res. Technol. Aqua 62, 417–425 (2013).
Chen, L., Zhang, J. J. & Zheng, X. L. Coupling technique for deep removal of manganese and iron from potable water. Environ. Eng. Sci. 33, 261–269 (2016).
Adam, L. C., Fabian, I., Suzuki, K. & Gordon, G. Hypochlorous acid decomposition in the pH 5–8 region. Inorg. Chem. 31, 3534–3541 (1992).
Farley, K. A. et al. Light and variable 37Cl/35Cl ratios in rocks from Gale crater, Mars: possible signature of perchlorate. Earth Planet. Sci. Lett. 438, 14–24 (2016).
Brundrett, M., Yan, W., Velazquez, M. C., Rao, B. & Jackson, W. A. Abiotic reduction of chlorate by Fe(ii) minerals: implications for occurrence and transformation of oxy-chlorine species on Earth and Mars. ACS Earth Space Chem. 3, 700–710 (2019).
Miller, L. G., Baesman, S. M., Carlström, C. I., Coates, J. D. & Oremland, R. S. Methane oxidation linked to chlorite dismutation. Front. Microbiol. 5, 275 (2014).
Zhang, X. et al. Reaction of methane and UV-activated perchlorate: relevance to heterogeneous loss of methane in the atmosphere of Mars. Icarus 376, 114832 (2022).
Smith, M. L., Claire, M. W., Catling, D. C. & Zahnle, K. J. The formation of sulfate, nitrate and perchlorate salts in the Martian atmosphere. Icarus 231, 51–64 (2014).
Catling, D. C. et al. Atmospheric origins of perchlorate on Mars and in the Atacama. J. Geophys. Res. Planets 115, E00E11 (2010).
Turner, A. M., Abplanalp, M. J. & Kaiser, R. I. Mechanistic studies on the radiolytic decomposition of perchlorates on the Martian surface. Astrophys. J. 820, 127 (2016).
Wu, Z. C. et al. Forming perchlorates on Mars through plasma chemistry during dust events. Earth Planet. Sci. Lett. 504, 94–105 (2018).
Carrier, B. L. & Kounaves, S. P. The origins of perchlorate in the Martian soil. Geophys. Res. Lett. 42, 3739–3745 (2015).
Melwani Daswani, M. & Kite, E. Paleohydrology on Mars constrained by mass balance and mineralogy of pre‐Amazonian sodium chloride lakes. J. Geophys. Res. Planets 122, 1802–1823 (2017).
Kim, Y. S., Wo, K. P., Maity, S., Atreya, S. K. & Kaiser, R. I. Radiation-induced formation of chlorine oxides and their potential role in the origin of Martian perchlorates. J. Am. Chem. Soc. 135, 4910–4913 (2013).
Kang, N., Anderson, T. A., Rao, B. & Jackson, W. A. Characteristics of perchlorate formation via photodissociation of aqueous chlorite. Environ. Chem. 6, 53–59 (2009).
Bethke, C. M. Geochemical and biogeochemical reaction modeling. (Cambridge University Press, 2007).
Catalano, J. G. Thermodynamic and mass balance constraints on iron-bearing phyllosilicate formation and alteration pathways on early Mars. J. Geophys. Res. Planets 118, 2124–2136 (2013).
Delany, J. & Lundeen, S. R. The LLNL Thermochemical Database Report UCRL-21658 (Lawrence Livermore National Laboratory, 1990).
Sternbeck, J. Kinetics of rhodochrosite crystal growth at 25 °C: the role of surface speciation. Geochim. Cosmochim. Acta 61, 785–793 (1997).
Vaniman, D. T. et al. Magnesium sulphate salts and the history of water on Mars. Nature 431, 663–665 (2004).
Fox-Powell, M. G., Hallsworth, J. E., Cousins, C. R. & Cockell, C. S. Ionic strength is a barrier to the habitability of Mars. Astrobiology 16, 427–442 (2016).
Gellert, R. et al. Alpha Particle X‐ray Spectrometer (APXS): results from Gusev crater and calibration report. J. Geophys. Res. Planets 111, E02S05 (2006).
Marion, G., Catling, D. & Kargel, J. Br/Cl partitioning in chloride minerals in the Burns formation on Mars. Icarus 200, 436–445 (2009).
Ilton, E. S., Post, J. E., Heaney, P. J., Ling, F. T. & Kerisit, S. N. XPS determination of Mn oxidation states in Mn (hydr) oxides. Appl. Surf. Sci. 366, 475–485 (2016).
Cerrato, J. M., Hochella Jr, M. F., Knocke, W. R., Dietrich, A. M. & Cromer, T. F. Use of XPS to identify the oxidation state of Mn in solid surfaces of filtration media oxide samples from drinking water treatment plants. Environ. Sci. Technol. 44, 5881–5886 (2010).
Acknowledgements
This research was funded by NASA Science Mission Directorate Future Investigators in NASA Earth and Space Science and Technology (FINESST) programme through award no. 80NSSC19K1521. J.G.C. was supported by the NASA Exobiology programme through award no. 80NSSC18K1292. G.J.L. was supported by the National Science Foundation Graduate Research Fellowship Program under grant no. DGE-1745038 and DGE-2139839. Discussions with B. Jolliff, R. Arvidson and J. Hurowitz improved this manuscript. P. Carpenter is thanked for assistance with XRD data collection and Rietveld refinements. R. Arvidson and A. Knight are thanked for assistance in VNIR data collection.
Author information
Authors and Affiliations
Contributions
K.M. and J.G.C. designed the study. K.M. conducted the experiments with assistance from E.L.M. G.L. contributed X-ray photoelectron spectroscopy analyses. J.G.C. and K.M. performed the thermodynamic and kinetic modeling. K.M. analyzed the results and wrote the original manuscript, with additional text provided by J.G. and further editing by G.L. and E.L.M. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Geoscience thanks Yasuhito Sekine and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Tamara Goldin, in collaboration with the Nature Geoscience team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Reaction of Martian basalt with water equilibrated with 0.5 bar CO2 and 0.03 bar O2.
(a) Relationship of dissolved CO2 and O2 concentrations to pH. (b) Mass fraction of mineral alteration productions.
Extended Data Fig. 2 Mineralogy of the solids produced by oxidation of dissolved Mn(II) by 10 mmol L−1 bromate.
XRD patterns of the solids produced by reaction of 10 mmol L−1 Mn(II) with 10 mmol L−1 bromate in 100 mmol L−1 magnesium chloride fluids. Patterns are visually offset for clarity. Diagnostic peaks are labeled and indicated with dashed lines. See Extended Data Table 4 for complete sample details.
Extended Data Fig. 3 Images of the solids produced by oxidation of dissolved Mn(II) by bromate.
Minerals precipitated following reaction in magnesium chloride (a-d) and magnesium sulfate (e and f) fluids.
Extended Data Fig. 4 Comparison of VNIR spectra of the manganese oxide mineral detected at Endeavor crater, Mars, and produced in Mn(II) oxidation experiments.
Solids produced from oxidation of dissolved Mn(II) by bromate in (a) magnesium chloride and (b) magnesium sulfate fluids. The labels indicate the initial pH of the samples. See Extended Data Table 5 for complete sample details.
Supplementary information
Supplementary Information
Supplementary Figs. 1–4, Tables 1–4 and Discussion.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mitra, K., Moreland, E.L., Ledingham, G.J. et al. Formation of manganese oxides on early Mars due to active halogen cycling. Nat. Geosci. 16, 133–139 (2023). https://doi.org/10.1038/s41561-022-01094-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-022-01094-y